Watching a cation bind to a molecule in real time
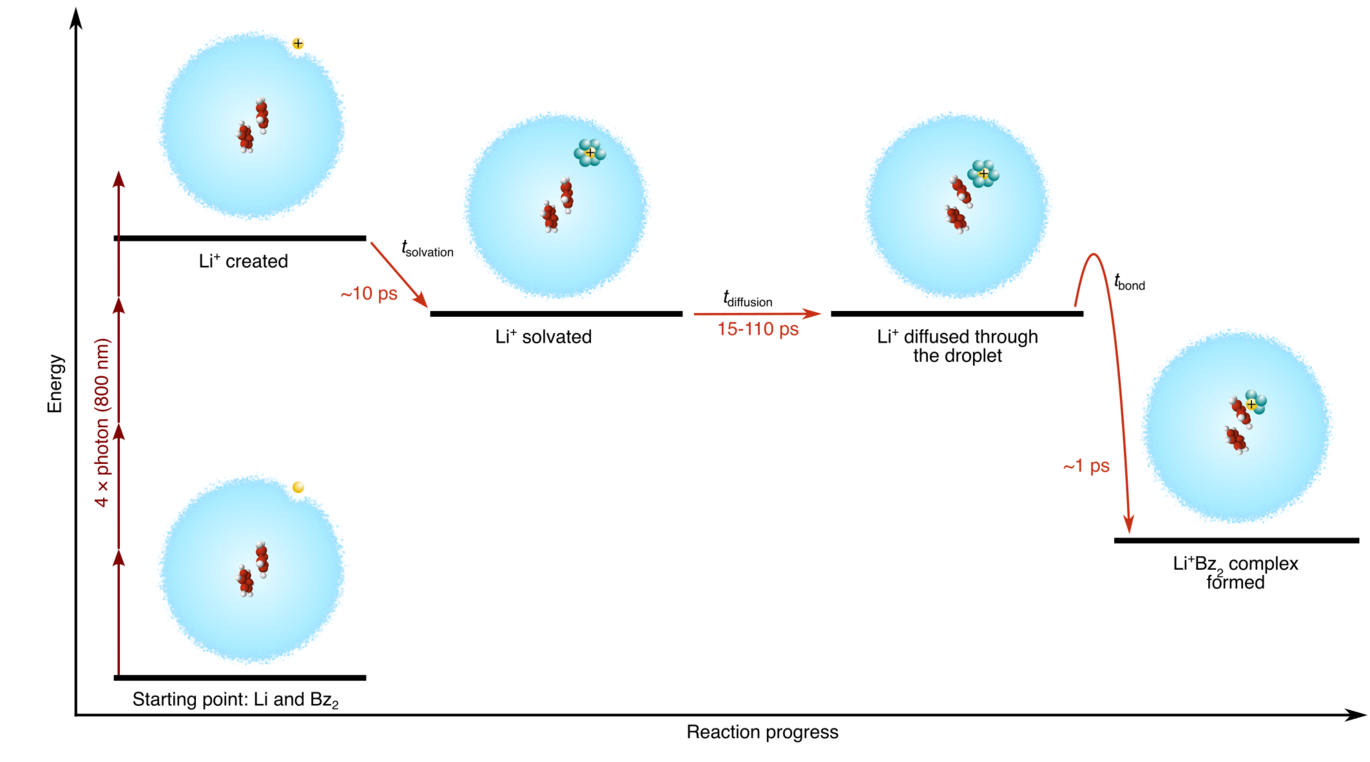
The decades-long dream of recording molecular movies – i.e. seeing how molecules change during chemical reactions on the natural femtosecond time scale – is currently finally coming true. So far, this happens almost exclusively for processes where chemical bonds are changed or broken by light. Bond-making processes, key to reactions between two molecules, remain elusive due to the difficulty of knowing when the reaction starts. For two atomic or molecular species to react, they must first meet. This happens by diffusion, a process that is not controllable on the femtosecond time. Therefore, the start of the reaction is ill-defined, which makes it difficult to shoot the molecular movie.
Now, Jeppe K. Christensen, Christian Engelbrecht Petersen, Simon. H. Albrechtsen, Jean Goudot, Florent Calvo and Henrik Stapelfeldt have introduced an experimental method that makes it possible to prepare two reactants, an ion and a molecule, at a controllable initial distance. The technique, recently published in Nature Communication and selected as an Editors Highlight, uses nanometer-sized droplets of liquid helium. In each droplet, a molecule, here benzene, is placed in the center and a lithium atom at the surface. To start the action, a femtosecond laser pulse irradiates the system and selectively ionizes the Li atom. This creates a Li+ ion at a well-defined time and at a well-defined distance to the benzene molecule. The initial solvation of Li+ in the helium solvent, its diffusion through the droplet and its final reaction with the benzene molecule to form an ion-molecule complex is monitored by irradiating the system with a second femtosecond laser pulse sent at a number of different times, thereby recording a molecular movie. The results show that the reaction is diffusion-limited and that the Li+ ion takes about 29 picoseconds to reach and bind to the benzene molecule for a droplet with a radius of 40 Å. This corresponds to a reaction rate constant which is more than 100 times higher than for reactions in conventional chemical solvents such as water – most likely a result of the superfluid properties of the helium droplet.
The method and findings open opportunities for imaging stereodynamics of chemical reactions in real-time, i.e. seeing how molecules rotate as they approach each other to enter the most favorable orientation for the reaction. Chemists take this key concept for granted in their description of reactions, but it has never been observed experimentally.
The work is financed by a Villum Investigator grant from the Villum Foundation.
Link to article: https://www.nature.com/articles/s41467-025-68008-5
Link to Editors Highlights: https://www.nature.com/collections/wtpqpqpgwd
